Metabolic Signatures of Acinetobacter baumannii and Klebsiella pneumoniae Infections in Acute-on-chronic Liver Failure
Abstract
Background & Aims
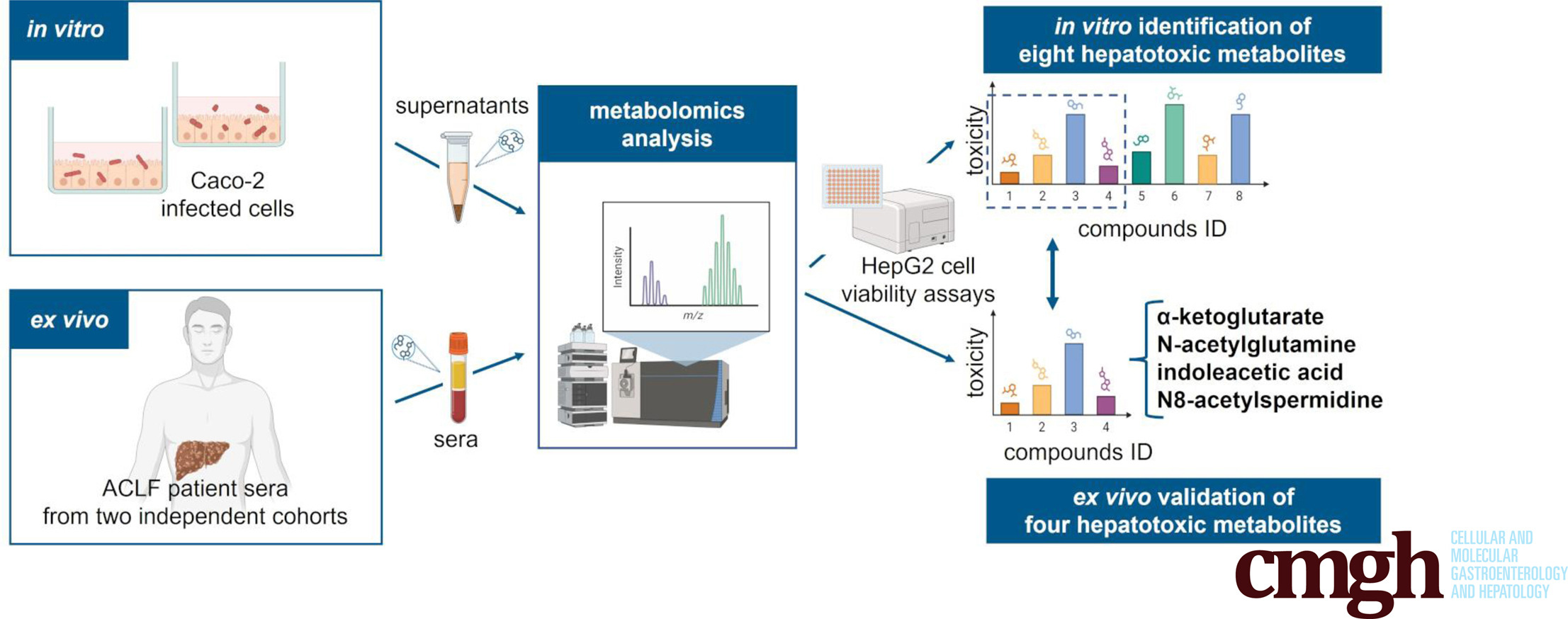
Acute-on-chronic liver failure (ACLF) is a life-threatening syndrome of acute hepatic decompensation (AD) that leads to multiorgan failure and high mortality. Bacterial infections are often implicated in ACLF pathogenesis; however, their underlying molecular mechanisms remain poorly understood. This study employed a combined in vitro-ex vivo metabolomics approach to investigate infection-associated metabolic alterations relevant to ACLF.
Methods
Gut (Caco-2) cells were infected with Acinetobacter baumannii and Klebsiella pneumoniae strains. Metabolite profiling was conducted on cell culture supernatants, and selected metabolites were tested for hepatotoxicity in vitro using liver (HepG2) cells. Metabolomic analysis of sera from 2 independent patient cohorts (AD and ACLF) was conducted to validate in vitro findings and to assess their clinical relevance.
Results
Distinct metabolic signatures were identified in Abaumannii (19 metabolites) and Kpneumoniae (15 metabolites)-infected Caco-2 cells. Four key metabolites from each bacterial species were prioritized for further experiments: α-ketoglutarate, indoleacetic acid, p-coumaric acid, uridine (Abaumannii), desthiobiotin, N8-acetylspermidine, N-acetylglutamine, and β-pinene (Kpneumoniae). Hepatotoxicity was demonstrated in liver (HepG2) cells exposed to Caco-2 infected cell-derived supernatants, infection-associated metabolites, and metabolite mixtures (in all conditions, P < .0001). Increased levels of α-ketoglutarate (P = .0002), N-acetylglutamine (P = .0153), indoleacetic acid (P < .05), and N8-acetylspermidine (P < .01) have been confirmed in the sera of patients with AD and ACLF.
Conclusions
Our findings suggest that metabolites associated with bacterial infections and hepatotoxic potential are significantly elevated in patients with AD and ACLF. These compounds may contribute to disease-related metabolic disturbances, representing promising candidates as early diagnostic biomarkers and targeted therapeutic strategies for ACLF.
